|
3/15/2024 0 Comments Bohr model of an oxygen atom 
For example, if two electrons are filled in the ‘s’ subshell of the first shell, the resulting notation is ‘1s 2’.These labels contain the shell number (given by the principal quantum number), the subshell name (given by the azimuthal quantum number) and the total number of electrons in the subshell in superscript.The electron configuration of an atom is written with the help of subshell labels.Thus, it can be understood that the 1p, 2d, and 3f orbitals do not exist because the value of the azimuthal quantum number is always less than that of the principal quantum number. Resulting Subshell in the Electron Configuration Therefore, the s, p, d, and f subshells can accommodate a maximum of 2, 6, 10, and 14 electrons, respectively.Īll the possible subshells for values of n up to 4 are tabulated below.The maximum number of electrons that can be accommodated by a subshell is given by the formula 2*(2l + 1).The subshells correspond to l=0, l=1, l=2, and l=3 and are named the s, p, d, and f subshells, respectively. Therefore, when n has a value of 4, four different subshells are possible. This quantum number is dependent on the value of the principal quantum number, n.The subshells into which electrons are distributed are based on the azimuthal quantum number (denoted by ‘l’). 
The shells, values of n, and the total number of electrons that can be accommodated are tabulated below. It is represented by the formula 2n 2, where ‘n’ is the shell number. The maximum number of electrons that can be accommodated in a shell is based on the principal quantum number (n). This notation for the distribution of electrons in the atomic orbitals of atoms came into practice shortly after the Bohr model of the atom was presented by Ernest Rutherford and Niels Bohr in the year 1913.Īlso Read ⇒ Interactive Periodic Table Writing Electron Configurations Shells Predicting the properties of a group of elements (elements with similar electron configurations tend to exhibit similar properties). 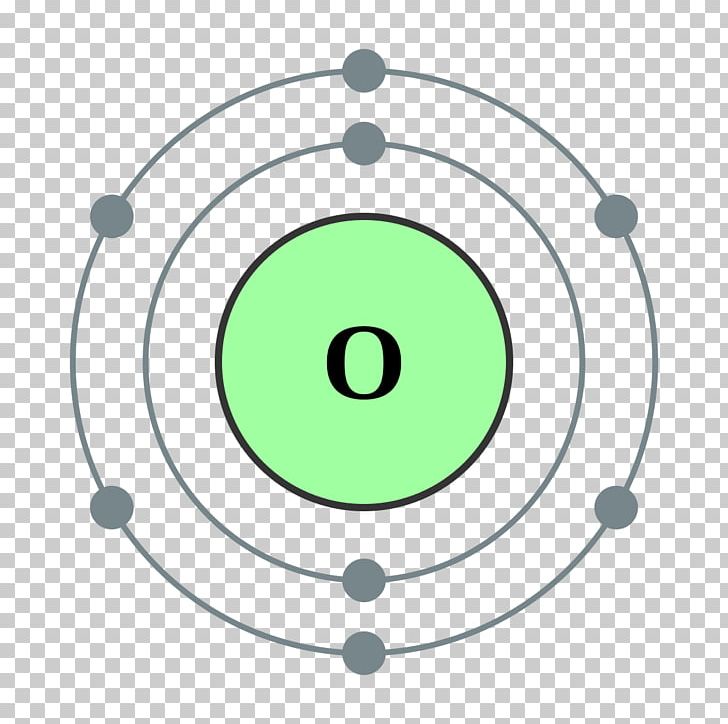
Therefore, the abbreviated electron configuration of sodium is 3s 1 (the electron configuration of neon is 1s 22s 22p 6, which can be abbreviated to 2s 22p 6). In the abbreviated notation, the sequence of completely filled subshells that correspond to the electronic configuration of a noble gas is replaced with the symbol of that noble gas in square brackets. In such cases, an abbreviated or condensed notation may be used instead of the standard notation. However, the standard notation often yields lengthy electron configurations (especially for elements having a relatively large atomic number).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
